We offer a range of veterinary courses depending on the areas a vet surgeon wants to specialize in, Learn more about our courses
Standing on the Shoulders of Giants
“Yes, Dr. Lillihei, Yes sir, it is an honour.” I was standing in the hardware store when I found myself speaking with the founding father of cardiac surgery. I had started my surgical residency at the Virginia Maryland Regional College of Veterinary Medicine about 3 months before. Most residencies require a research project. My father’s career had spurred my interest in cardiac surgery. My residency supervisor also had an interest in cardiac surgery, and specifically, the adaptation of a procedure which had been done in humans 40 years before. In this procedure, the parent was used as a heart lung machine to operate on their child with a congenital heart defect. It was called controlled cross-circulation.
Controlled cross-circulation was developed by Dr. C. Walton Lillihei in 1952. He had originally invented it to allow training cardiac surgeons experience working inside a beating heart. What he did not expect was that it was much gentler to platelets and red blood cells than the heart lung machines of the day. He operated on laboratory dogs using this technique and found them the morning after surgery wagging their tails and ready to eat. He had the crazy idea of using this technique in humans.
The first open heart surgery ever performed was done by Dr. Lillihei in September 1952 in a child with a congenital heart defect. He induced profound hypothermia by submerging the child in an ice bath. This allowed him to stop the heart and drain it of blood with enough time to repair the hole in the child’s atrial septum. Several patients were operated with this technique with good results. Surgeons, however, quickly recognised the limitation of hypothermic cardiac surgery which was that the brain and other organs could only survive for a limited period of time without circulation.
This spurred early cardiac surgeons to investigate cardiopulmonary bypass. The primary function of cardiopulmonary bypass is to direct blood away from the heart allowing an unobstructed view. The secondary function is to protect the heart and other organs during the procedure. The method of cardiopulmonary bypass that held the most promise in the early1950’s was using a bubble oxygenator. Blood being drained from the heart was bubbled with oxygen creating a foam. The blood was defoamed and then pumped into the aorta under pressure. It carried the risk of stroke due to injection of air bubbles. It was also very damaging to red blood cells and platelets. In 1953 the first successful open heart surgery was reported using cardiopulmonary bypass via a bubble oxygenator. Its pioneer abandoned the technique because of a series of subsequent failures.
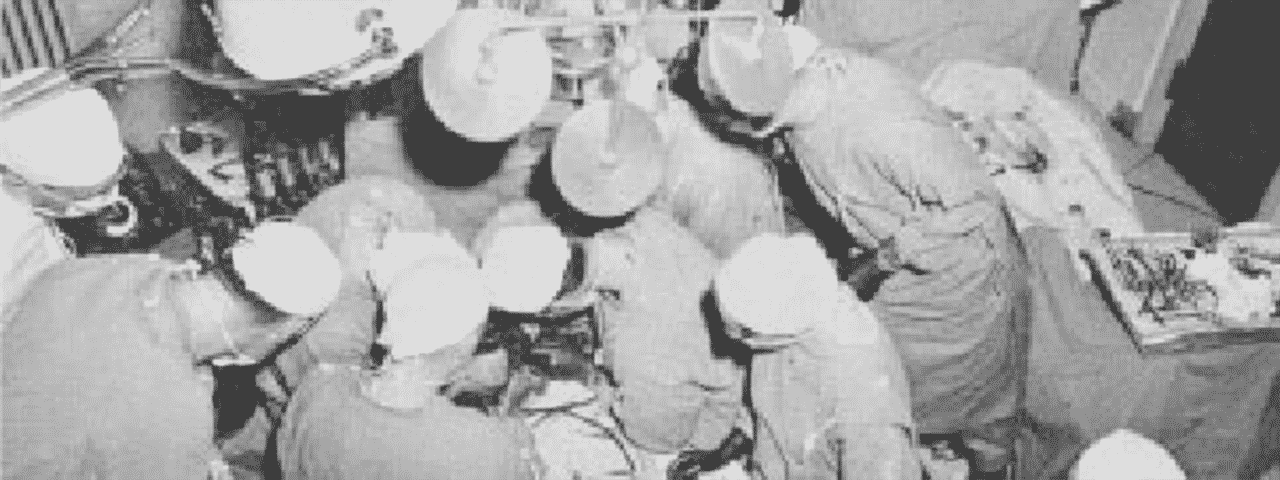
During this period, Dr. Lillihei was perfecting the technique of controlled cross circulation on laboratory dogs. In March 1954, he operated on the first human using this technique. This first patient, a 9 year old boy with a ventricular septal defect, died of an infection 9 days later, but the potential of this technique was recognised. Dr. Lillihei operated on 45 children with congenital heart defects over the next 15 months using this technique with a 66% 30 year survival rate. Controlled cross circulation was abandoned in July 1955 with critics citing the fact that this was the only surgery with a potential for a 200% mortality. Dr. Lillihei is viewed as the founding father of open heart surgery and continued to pioneer new techniques over the next several years.
First human ever operated using Controlled Cross-Circulation
My resident supervisor’s vision was to use greyhounds as the heart-lung machine for dogs with cardiac abnormalities. The owners of the patient would be required to adopt the greyhound following the surgery. Bubble oxygenator cardiac surgery had not been particularly successful in dogs because their red blood cells particularly fragile and easily damaged. Membrane oxygenators had since replaced bubble oxygenators in humans because they were gentler on the red blood cells and carried a much lower risk of stroke. At about $3000 USD each, they were, however, prohibitively expensive for use in dogs.
I called my Dad, a cardiac surgeon himself, to let him know that I was looking at reintroducing controlled cross circulation for dogs with structural cardiac abnormalities. He said “That was Lillihei, wasn’t it?” “Yes” I replied. He said “He’s still alive. I think he lives in Minnesota.” I went to the library and found him in Who’s Who in American Physicians as well as his address. I called telephone information in Minnesota and got his home phone number. I called him and left a message.
Back to the hardware store… I received a message on my old-fashioned voice pager. Dr. Lilliehei was on the phone. This was the beginning of an improbable friendship that lasted years. He invited me to his laboratory in Minnesota where I spent 3 weeks observing laboratory animal and human cardiac surgery. He also enthusiastically agreed to be on my Master’s committee. He travelled to Blacksburg to attend the defence of my thesis. Just before my presentation, he did a short lecture called “The history of cardiac surgery.” He showed video of the world’s first open heart surgeries. I will never forget the look on the parent’s face of a little girl with a devastating cardiac defect called Tetraology of Fallot. Her lips were blue because of the unoxygenated blood which was circulating through her body. Her parents were terrified as they handed her over to Dr. Lillihei on that day in 1954. Tetrology of Fallot had never been repaired before. To my knowledge, she is still alive today.
“So, how would you like to remove this one?” our internal medicine specialist asked jokingly as he showed me an ultrasound picture of a golfball sized tumour filling the entire right atrium of a dog. Willow, and 8 year old Rottweiler, presented with an abdomen full of fluid. The fluid had developed over the preceding months when the tumour in her atrium had blocked the blood return to the heart. She had a very distended belly but was otherwise quite happy and very beautiful. Never one to shy from a challenge, I said “sure.” I had just started working with this specialist and he did not expect me to say yes.
With a few days of planning, I was ready to proceed. I did a right lateral thoracotomy which means that I made an incision between Willow’s 4th and 5th ribs. Immediately upon opening the chest, I could see the right atrium and vena cavae bulging, distended with blood. I placed a circular suture in the tip of the right auricle which is an appendage on the right atrium. I made an incision in the middle of the suture and introduced my index finger into the heart. With the tip of my finger I could feel the attachment of the tumour to the atrial septum which is the wall that divides the right and left atria. It was the first time I had my finger inside the beating heart of a clinical patient. It was kind of exciting.
I preplaced sutures in two parallel lines in the wall of the right atrium. Each suture was clamped with a haemostat. When lifted, the two lines of sutures looked like the lines on a parachute. I then placed temporary ligatures around the vena cavae and the azygous vein. With all three of these vessels tied off, almost all of the bloodflow to the right atrium was interrupted. This gave me the chance I needed. I called for the anaesthetist to start the timer and I swiftly made an incision between the two rows of sutures that I had previously placed. The tumour came into view. Another first, I had never seen the inside of a patient’s beating heart. While I did not have time to relish the moment, I was quite excited. I grabbed the tumour in my left hand and cut it off at its base, being careful not to cut open the atrial septum. I only had about 2 minutes to complete the part inside the heart. I lifted up the preplaced sutures in the atrial wall and put a Satinsky clamp across the base. I carefully released the clamp a bit and removed the ligature from the vena cava. This caused blood to well up in the atrium and to force any bubbles out of the heart. Again, I closed the Satinsky. I closed the incision in the right atrium and I was home free. Willow recovered uneventfully and urinated out 10 litres of abdominal fluid over the next 24 hours. She was a new dog.
I never fulfilled my dream of being a veterinary cardiac surgeon other than a handful of clinical cases over the years. Without a fully-staffed ICU, cardiopulmonary bypass surgery is impossible. Technically, the surgeries are not too difficult. The aftercare is where the patients live or die. There are a couple of surgeons at our hospital who are interested in starting a cardiac surgery unit and I am very happy to serve as a mentor and to add my very outdated knowledge. While I will probably never be a clinical cardiac surgeon, these cases will always have a special place in my heart.